.
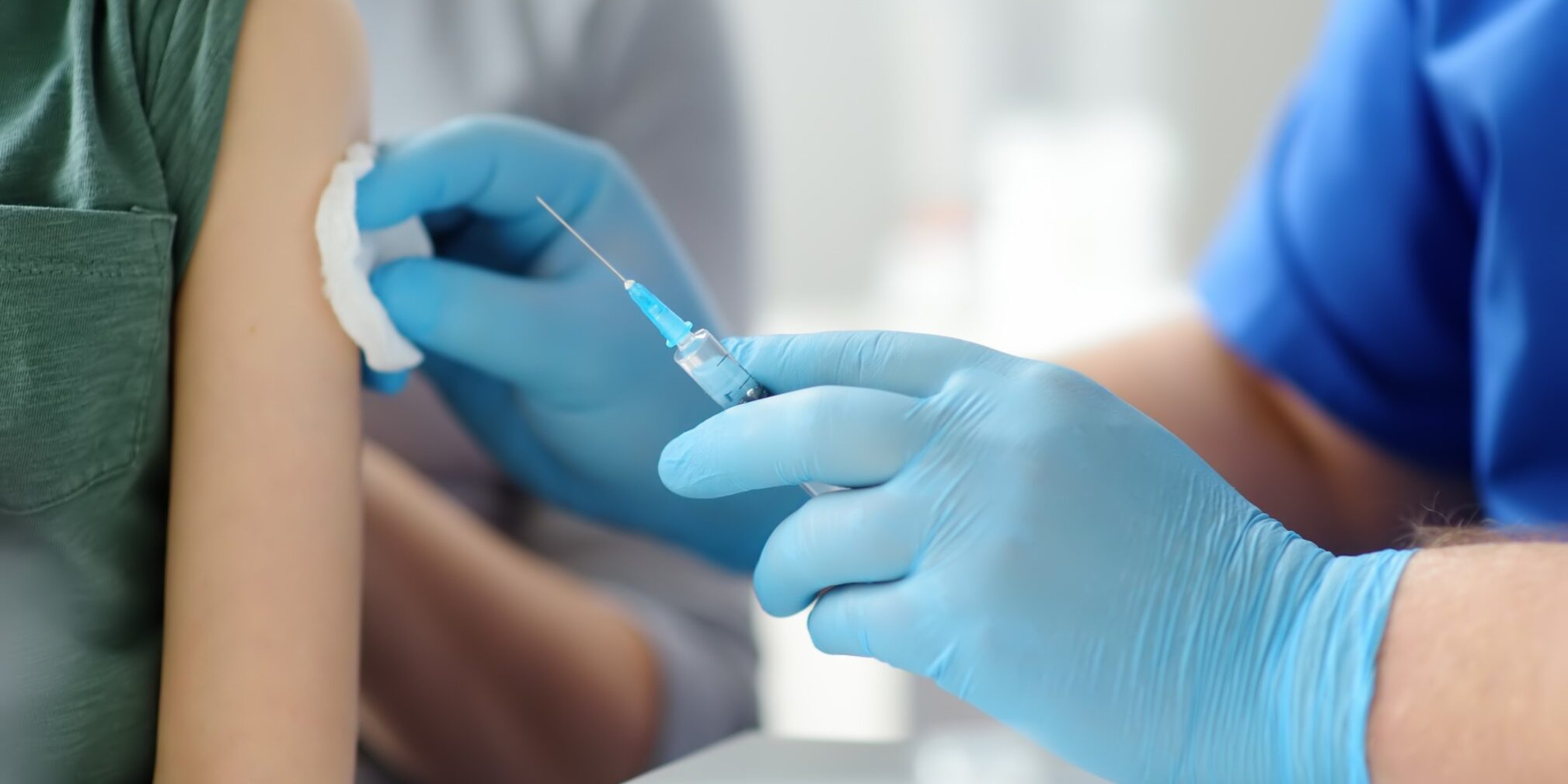
With the outbreak of the Covid -19 pandemic, the world was caught unawares on how to combat the disease. There were no preventive vaccines available that people who got infected with the disease could take so as to reduce the intensity of the disease on those infected. Research institutions and organizations like Docsun Biomed,Pfizer,Johnson and Johnson and others under the world health organizations were called upon to come up with vaccines and devises that could be used in the fight against further spread of the covid-19 disease. The calls for the experts in the healthcare industry to come up with vaccines piqued interest in knowing what entails the development of a vaccine before it is deemed safe to be used in the general population.
THE SCIENCE PHASE.
This is where a lot of research is conducted against a particular disease and explored on the response it elicits on one’s immunity. A lot of investment is pooled together so as to speed up the process especially when it comes to a disease like the covid-19 virus.
TRIAL PHASE
This is a phase where the vaccine samples are introduced to a select number of humans and their response is monitored. The data collected on this phase gives the experts a clear understanding on the effectiveness against the disease any side effects on any of the participants of the trail. The trials on the people are taken under very controlled settings by the healthcare experts.
ASSESSMENT STAGE.
This is the phase where the potential vaccine is given to a large group of people. They are given the vaccine samples under normal natural conditions of when the disease occurs. This is said to be a real-life assessment of the efficacy of the disease
THE PRODUCTION PHASE.
Once the sample vaccine has undergone all the phases and declared fit for use, the product has to further get approval by regulators in that specific sector. The safety of the product has to be paramount to make sure no accidents happen. Once this is cleared the manufacturing of the vaccine is given the go ahead and the vaccine is made available for use.
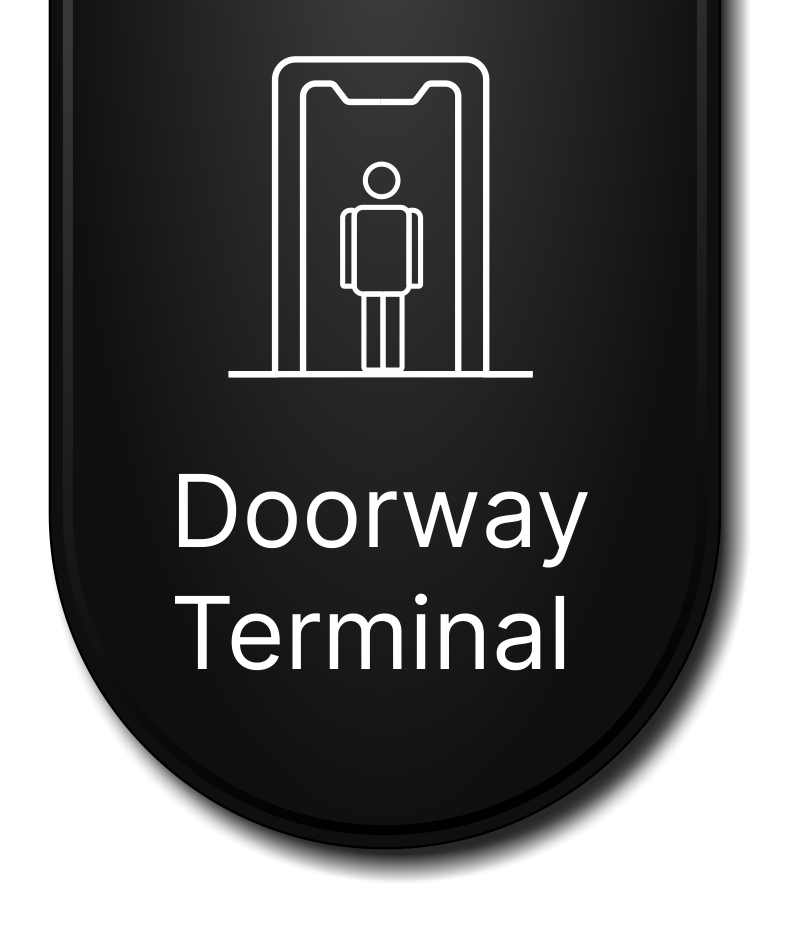
As for the covid-19 vaccine a lot of research is still ongoing even with the release of the current Covid-19 vaccines. People are also encouraged to get tested as new variants of the covid-19 are on the rise. The data collected on these will go a long way in helping healthcare experts improve the efficacy of future vaccines. Docsun Biomed has come up with several new devices that aid in the non-invasive mode of Covid-19 testing and monitoring using the OCT technology. Some of their devises include the Docsun web platform, the Docsun mobile app and the Docsun doorway terminal.